- Home Page
- Company Profile
-
Our Products
- Dyes Intermediates
- Dyes and Dye Intermediates
- Pyrazolones Dye Intermediates
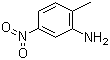
- BETA NAPTHOL (2 NAPTHOL)
- Dipehenylamine
- 1 NAPTHOL 4 SULPHONIC ACID
- 1 NAPHTHYL AMINE 6: SULPHONIC ACID
- 1 NAPHTHYL AMINE 7: SULPHONIC ACID
- BRONNER ACID
- 1,2,4 DIAZO Acid
- 1,3 Spmap
- 1,3 SPMP -3-methyl-5-pyrazolone
- 1,3 SPMP, 1-(3'-Sulfophenyl)-3-methyl-5-pyrazolone
- 1,6 Cleves Acid
- 1,7 cleves Acid
- 1-4 SPCP , 1-(4-Sulfophenyl)-3-carboxy-5-pyrazolo
- 1-(4-Sulfophenyl)-3-carboxy-5-pyrazolo Beige powder
- 2 NADPSA
- 2,5 DCA
- 2,5 DCSA
- 2-Chloro PPD Sulphate
- 2-Pyridone
- 2R Acid , Sulpho Gamma Acid
- 4 Adapsa
- 4 CAP
- 4 CAPSA
- 4 Chloro Metanilic Acid
- 4 NADPSA
- 4 NAP -4- Nitro 2- Amino Phenol
- 4 NAPSA
- 4,4 DABA- 4,4'-Diamino Benzanilide
- 5 NAP, 5 NItro 2 Amino Phenol
- 6 NAPSA
- 6 Nitro 1,2,4 diazo acid
- 6-ACETYL OAPSA
- Acetyl Bronners acid
- Acetyl Gamma Acid
- Liquid Acrylic acid
- Acetyl MPDSA
- Amido Epsilon Acid
- Amido G Acid
- Aniline 2,4 Disulphonic Acid
- Aniline 2,5 Disulphonic acid
- Anthraquinone
- Armstrong Acid
- BDSA -Bezedine 2,2 Di sulfonic acid
- Grey Fine Crystals Blue B Base
- BNSA, Naphthalene-2-sulfonic acid
- BON Acid 92-70-6
- BRONNER VS
- C ACID
- Chloranil
- Cyanuric Chloride
- DASA
- DASDA
- DCSPMP
- Di J ACID
- DMAVS Black Powder
- DMAVS
- EBAMSA
- Epsilon Acid
- EUROPEAN K ACID
- F C ACID
- G Salt
- Gamma Acid
- H Acid
- J acid
- K Acid
- KOCH Acid
- Laurent Acid
- Leuco quinizarin
- MAA-Methyl Acetoacetate CAS No 105-45-3
- MAP-Meta Amino Phenol
- Metanilic Acid
- METHYL OAPSAMIDE
- Light Yellow Liquid Monoethylamine
- MPD-meta-Phenylenediamine
- MPDDSA
- MPDSA
- MSPG
- MUA, Meta Ureido Aniline
- MXOSA
- N METHYL J ACID
- N Phenyl J Acid
- N W ACID
- N-Phenyldiethanolamine
- NAS-5
- o-Anisidine
- OAPSA
- OAPSAMIDE
- OAVS
- OBSA, 2-formyl-Benzenesulfonic acid
- OCA5SA
- OCPNA
- ONAPSA
- ONCBSA
- Ortho Toludine
- OT Base
- OT5SA
- p-Anisidine
- p-Toluidine
- PAA-Para Amino Acetanilide
- PAAB 3,4 DSA
- PAABSA- Para-Aminoazobenzene-4-sulfonic acid
- PCONA
- PCVS
- PNAOSA
- PNCBOSA
- PNTOSA
- PPDOSA
- PTOSA
- PTPMP
- Quinizarin
- R Salt
- Resist Salt, Meta Nitrobenzene Sulphonic Acid
- Resorcinol Chemical
- Salicylic Acid CAS No 69-72-7
- Schaeffers Acid
- Sodium Napthionate
- Sodium Sulphide 60% YELLOW FLAKES Fe<20 PPM
- Sulphanilic acid
- Sulpho C Acid
- Sulpho Tobias Acid Dye
- Sulpho Tobias Acid
- Sulpho VS
- Tobias Acid Dye Intermediates
- Vinyl Sulphone Ester
- Violet Acid
- Para Toluidine
- Crude Naphthalene
- Ethyl Cyanoacetate
- Crystal Resorcinol
- Ethyl Cynoacetate
- Venyl Acetate Monomer
- Pigments Intermediates
- Pigment Intermediates
- 1,3,5 PMP - 1-Phenyl-3-methyl-5-pyrazolone
- 2,4 DNA
- 2,4,5 TCA
- 2B acid
- 3,3 DCB - 3,3-Dichloro Benzidine
- 4B Acid
- Liquid Aluminium chloride
- Anilide AAOCA AcetoAcet-O-Chloroanilide
- Anilide AAA Acetoacetanilide
- Anilide AAMX Acetoacetic acid m-xylidide
- Anilide AAOA o-Acetoacetanisidide
- Anilide AAOT o-Acetoacetotoluidide
- Blue B Base
- BON Acid
- CLT ACID
- Fast Red KD Base
- Fast Scarlet G Base CAS No 99-55-8
- Gum Rosin WW
- MNPT
- Naphthol AS
- Naphthol AS-D
- Naphthol AS-E
- Naphthol AS-IRG
- Naphthol AS-OL
- Naphthol AS-PH
- Naphthol ASG
- P-T-M-P
- Para Amino Benzamide
- Sodium Nitrite
- Pharma Intermediates
- Food Colours
- Intermediate, Chemicals & Solvents
- Vinyl Sulphone Base Dye
- Fast Basis
- Bifunctional Reactive Dyes
- Direct Dyes
- Naphthol Dyes
- Acid Dyes
- Solvent Dyes
- Reactive Dyes
- Acrylamide
- Acrylic acid
- Alpha Naphthol
- Aniline
- Beta Naphthol
- Caustic Potash
- DCDA Dicyandiamide
- DEA-Diethanolamine
- DMF-N,N-Dimethylformamide
- DPA-Diphenylamine
- ECH-Epichlorohydrine
- EDA-Ethylene Diamine
- EDTA 2Na
- EDTA 4Na
- EDTA pure acid
- Ethyl acetoacetate
- Vat Brown G A/C
- Organic Pigments
- Pigment Red 48:2
- Pigment Blue 15:0
- Pigment Blue 15:3
- Pigment Blue 15:3 A
- Pigment Green 7
- Pigment Orange 34
- Pigment Orange 34 A
- Pigment Orange 5
- Pigment Orange 5 A
- Pigment Red 112
- Pigment Red 112 A
- Pigment Red 122
- Pigment Red 146
- Pigment Red 170
- Pigment Red 170 A
- Pigment Red 3
- Pigment Red 4
- Pigment Red 5
- Pigment Red 48:3
- Pigment Red 48:4
- Pigment Red 53:1
- Pigment Red 57:1
- Pigment Red 63:1
- Pigment Red 8
- Pigment Violet 23
- Pigment Yellow 1
- Pigment Yellow 13
- Pigment Yellow 14
- Pigment Yellow 17
- Pigment Yellow 3
- Pigment Yellow 62
- Pigment Yellow 65
- Pigment Yellow 74
- Pigment Yellow 74 A
- Pigment Yellow 83
- Pigment Yellow 83 A
- Inorganic Pigment
- Solvent Dyes
- Black RBL
- Black RE
- Black RL A
- Blue 2 GLN
- Fire Red
- Fire Red GLS
- Nigrosine Fat Soluble Induline Base
- Nigrosine Spirirt Soluble
- Nigrosine Spirirt Soluble Black
- Oil Blue M B A
- Oil Blue MIP
- Oil Lemon Yellow
- Oil Orange
- Oil Orange DXN
- Oil Orange ES
- Oil Red R
- Oil Scarlet Y Enquire Now
- Oil Yellow AM
- Oil Yellow DE
- Oil Yellow OAP Enquire Now
- Orange 2R
- Orange R
- Orange RE
- Orange RL
- Orange RL Powder
- Pink 5BLG
- Quinizarine Green SS.
- Red B
- Red BB A Solvent Dye
- Red BB A
- Solvent Blue 38
- Solvent Green 7 Dye Powder
- Solvent Green 7
- Victiria Blue B Basea
- Yellow 2RLS
- Yellow GL A
- Yellow R
- Basic Dyes
- Direct Dyes
- DIRECT CHRYSOPHENINE G
- DIRECT PAPER YELLOW 3GX
- DIRECT PAPER YELLOW T
- DIRECT SUN YELLOW RCH
- DIRECT TITAN YELLOW
- DIRECT YELLOW 2RLX
- DIRECT YELLOW 3GX
- DIRECT YELLOW 3RL
- DIRECT YELLOW 5GLL
- DIRECT YELLOW ARLX
- DIRECT YELLOW CFG
- DIRECT YELLOW G
- DIRECT YELLOW NNX
- DIRECT YELLOW NRS
- DIRECT YELLOW PG
- DIRECT YELLOW RLL
- DIRECT YELLOW RR
- DIRECT ORANGE 3R
- DIRECT ORANGE 7GLL
- DIRECT ORANGE GLL
- DIRECT ORANGE SE
- DIRECT ORANGE TGLL
- DIRECT ORANGE WS
- DIRECT VISCOSE ORANGE A
- DIRECT BRILLIANT FAST HELIO B
- DIRECT BRILLIANT VIOLET NB
- DIRECT BRILLIANTFAST VIOLET 2R
- DIRECT PAPER VIOLET BL
- DIRECT VIOLET 4BLL
- DIRECT VIOLET NBX
- DIRECT BLUE 2GDX
- DIRECT BLUE 2RLL
- DIRECT BLUE NB
- DIRECT COPPER BLUE 2R
- DIRECT COPPER BLUE GL
- DIRECT COTTON BLUE G
- DIRECT EVANS BLUE
- DIRECT LIGHT BLUE 5GLL
- DIRECT LIGHT FAST BLUE GL
- DIRECT SKY BLUE FB
- DIRECT SKY BLUE FF
- DIRECT SUPRA BLUE BRL
- DIRECT TRYPAN BLUE
- DIRECT TURQ BLUE FBL
- DIRECT TURQ BLUE GL
- DIRECT BENZOPURPURINE 4B
- DIRECT BORDEAUX 6B
- DIRECT BRILLIANT RED 8A
- Acid Dyes
- ACID FLORCSCEINE URANINE
- ACID LIGHT YELLOW 2GX
- ACID METANIL YELLOW MS
- ACID YELLOW 3GX
- ACID MILING FAST YELLOW MR
- ACID TARTARZINE ARX
- ACID YELLOW 2RX
- ACID YELLOW 3GL
- ACID YELLOW 5GN
- ACID YELLOW A4R
- ACID YELLOW FGX
- ACID YELLOW GLX
- ACID YELLOW M3RL
- ACID YELLOW M5RL
- ACID YELLOW NWX
- ACID YELLOW PGR
- ACID FUR YELLOW AEX
- ACID ORANGE 11
- ACID ORANGE 2R
- ACID ORANGE 2RL
- ACID ORANGE 3GX
- ACID ORANGE G
- ACID ORANGE GRX
- ACID ORANGE MRL
- ACID ORANGE N3RL
- ACID ORANGE RGN
- ACID ORANGE RL
- ACID ORANGE SG
- ACID YELLOW RL
- ACID BORDEAUX MB
- ACID BRILLIANT RED 10B
- ACID VIOLET 4BS
- ACID VIOLET 6B
- ACID VIOLET CARMINE 4B
- ACID VIOLET RL
- ACID VIOLET RLX
- LETHER VIOLET R
- ACID BORDEAUX RLX
- ACID BRILLIANT RED 2B
- ACID BRILLIANT RED 4G
- ACID BRILLIANT RED BL
- ACID CARMOISINE APX
- ACID FAST RED A
- ACID FUCHSINE SX
- ACID MAROON V
- ACID MILLING RED 3BN
- ACID MILLING RED RSNB
- ACID MILLING SCARLET G
- ACID PINK B
- ACID RED 2BX
- ACID RED 3GX
- ACID RED BROWN NS
- ACID RED BRX
- ACID RED BRXN
- ACID RED C5R
- ACID RED F2R
- ACID RED GN
- ACID RED GR
- ACID RED NB
- ACID RED 141
- ACID RED 1
- ACID RED 1 Powder
- ACID RUBINE 3GX
- ACID SCARLET 3R
- ACID BLUE 2G
- ACID BLUE A2GX
- ACID BLUE ANX
- ACID BLUE BRLL
- ACID BLUE MTR Reactive Dye
- ACID BLUE NBL
- ACID BRILLIANT BLUE FCF
- ACID BRILLIANT BLUE FF
- ACID FAST BLUE S5R
- ACID NAVY BLUE R
- ACID PETANT BLUE AS
- ACID PETANT BLUE VS
- C.I. GENERIC NAME ACID BLUE 1
- ACID BLUE 185
- ACID CYANINE GREEN G
- ACID DARK GREEN B
- ACID GREEN BLN
- ACID GREEN BS
- ACID BLUE MTR
- ACID GREEN UG
- ACID LEATHER GREEN NX
- ACID LEATHER GREEN WEG
- ACID MILLING GREEN 6B
- ACID OLIVE GREEN BGL
- ACID VAPORID GREEN PXX
- ACID BLACK 10BX
- ACID BLACK BGLX
- ACID BLACK LDN
- ACID BLACK MSRL
- ACID BLACK MT
- ACID BLACK SBX
- ACID BLACK WA
- ACID FAST BLACK 2BX
- ACID GREY APX
- ACID GREY AWW
- ACID GREY CG
- ACID GREY GLA Powder
- ACID GREY GLA
- ACID GREY RL Powder
- ACID GREY RL
- ACID LEATHER BLACK CB
- ACID MILLING BLACK NB
- ACID BROWN 2R
- ACID BROWN 2RL
- ACID BROWN 4BX
- ACID BROWN 5GX
- ACID BROWN CGX
- ACID BROWN CR
- ACID BROWN CTX
- ACID BROWN DS
- ACID BROWN GBX
- ACID BROWN GRX
- ACID BROWN GSX
- ACID BROWN HHK
- ACID BROWN M2RL
- ACID BROWN MBLX
- ACID BROWN MFX
- ACID BROWN NTX
- ACID BROWN ORG
- ACID BROWN PRG
- ACID BROWN RD
- ACID BROWN RGX
- ACID BROWN RX
- ACID BROWN S3RX
- ACID BROWN SGL
- ACID BROWN SRX
- ACID BROWN WG
- ACID BROWN WR
- ACID CAMEL BROWN APX
- ACID DARK BROWN 5R
- ACID DARK BROWN JBX
- ACID HAVANA BROWN 2RLX
- BEIGE APR
- BEIGE APX
- BROWN 2R
- BROWN DK
- BROWN DR
- BROWN GNX
- BROWN LN2RL
- BROWN P3R
- BROWN PHL
- BROWN RLL
- BROWN SCG
- BROWN SRLL
- BROWN TPN
- BROWN TPN 283
- BROWN XNP
- DARK BROWN
- DARK BROWN Acid 440
- DARK BROWN R
- OLIVE BROWN G
- Reactive Dyes
- Blue M4GD
- Blue MR
- G. Yellow MR
- Magenta MB
- Orange M2R
- Reactive Blue M2R
- Red M5B
- Red M8B
- Yellow M3R
- Yellow M4R
- Yellow M8G
- Black PN
- Blue P5G
- Blue P5RH
- Blue PGR
- G. Yellow PR
- Magenta PB
- Orange P2R
- Purple P3R
- Reactive Blue P3R
- Yellow M4G
- Red P2B
- Red P3B
- Red P6B
- Red P8B
- T. Blue PA
- Yellow P4G
- Yellow P8G
- Goldern Yellow MERL
- N Blue ME2R
- N. Blue ME2G
- Orange ME2R
- Reactive Blue HEGN
- Reactive Blue ME2RL
- Reactive Red ME3B
- Red ME4B
- Red ME6B
- Red MERB
- Supra Red 3BS
- Supra Yellow 3RS
- Violet ME2R
- Yellow ME3G
- Yellow ME4G
- Blue HERD
- G. Yellow HER
- Green HE4B
- N. Blue HE2R
- N. Blue HER
- Orange HE2R
- Orange HER
- Red HE3B
- Red HE7B
- Red HE8B
- Yellow HE4G
- Yellow HE4R
- Yellow HE6G
- Black B
- Blue BRF
- Blue R
- Brown GR
- G Orange 3R
- G Yellow R
- G. Yellow RNL
- Green 6B
- N Blue BF
- N Blue HRL
- N Blue RGB
- Orange 3R
- Reactive Black CNN VINYL SULPHONE DYES
- Reactive Black CNN
- Reactive Black GR
- Reactive Black GWF
- Reactive Black KGN
- Reactive Black R
- Reactive Black RL
- Reactive Black WNN
- Reactive Blue 3R
- Reactive Blue GG
- Red 3B
- Reactive Red GF
- Red 5B
- Red BB
- Red BS to ID
- Red F3B
- Red RB
- Red RBL
- VINYL SULPHONE DYES Red RBL
- Royal Blue BB
- Scarelt 2GF
- T Blue G
- Violet 5R
- Yellow FG
- Yellow GL
- Yellow GR
- Black PD**
- Blue BRF REACTIVE Blue 221
- Black PD2R**
- Black WNN*
- Dark Blue PDF*
- Navy Black
- Orange PD2R
- Reactive Black RGB
- Reactive Blue RGB
- Reactive Blue FNG
- Reactive Blue RGB Dyes
- Reactive Blue RR
- REACTIVE CARMINE RGB
- REACTIVE OCEAN BLUE SR
- Reactive Red 222
- Reactive Red RGB
- Reactive Yellow RGB
- Reactive Yellow RR
- Red PD3B
- Red PD3Y
- Red RR
- Yellow PD
- Yellow PD2G Dye
- Yellow PD2G
- Chemicals & Intermediates
- Aceto Acetic Methyl Easter
- Aceto Acetic Ethyal Easter
- N N Dimethylaniline
- N. N. Diethylaniline
- N. N. Diethylaniline Powder
- Ortho Toluidine
- Refined Naphthalene
- Para Toludine
- Crude Naphalene
- Mythyl Cynoacetate
- Ortho Anisidine
- Para Anisidine
- Sulpanilic Acid
- Sodium Naphthionate 74%
- Aceto Acetanilide
- Para Cresol
- Oro Cresol
- Oro Cresol Powder
- Diemino Stilbene Disulphonic Acid
- Mono N Buylamine
- Salicilic Acid
- Mono Eythlamine 70%
- Pure Di Ethanolamines
- Di Ethanolamines
- White Metanilic Acid Powder
- Aniline Oil
- Tobias Acid
- N. W. Acid 65%
- Meta Phenylene Diamine
- Meta Nitro Aniline
- Di N Buylamine
- Schaeffer's Acid
- Acetone
- Methanol
- Ethanol
- N-Buatanol
- IsoPropyl Alcohol
- Phenol
- Methyl meta Acrylate
- O P DIchlorobenzene
- Methylene Dichloride
- Dimethyl Formamide
- DimethylFormamide
- Methyl Acrylate
- Xylenes
- Benzene
- Acetic Acid
- Butyl Acrylate Monomer
- Epichlorohydrine
- Color Pigments
- Pigment Emulsifier
- Dyes Intermediates
- Contact Us
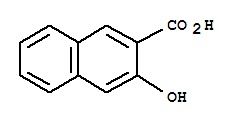
2-Hydroxy 3-Carboxy Naphthalene
500.00 - 5000.00 INR/Kilograms
Product Details:
- Storage Room Temperature
- Melting Point 142-146 degree centigrade
- Boiling point 238 - 239 degree centigrade
- Smell Fragrant
- Taste Odorless
- Grade Medicine Grade
- Physical Form Liquid
- Click to view more
X
2-Hydroxy 3-Carboxy Naphthalene Price And Quantity
- 1 Kilograms
- 500.00 - 5000.00 INR/Kilograms
2-Hydroxy 3-Carboxy Naphthalene Product Specifications
- Odorless
- 238 - 239 degree centigrade
- Medicine Grade
- Room Temperature
- Fragrant
- 142-146 degree centigrade
- Liquid
2-Hydroxy 3-Carboxy Naphthalene Trade Information
- Cash Against Delivery (CAD) Cash on Delivery (COD) Letter of Credit (L/C) Western Union Paypal Letter of Credit at Sight (Sight L/C) Telegraphic Transfer (T/T) Delivery Point (DP) Days after Acceptance (DA) Cash in Advance (CID) Cheque Cash Advance (CA)
- 50 Kilograms Per Day
- 1- 3 Days
- Yes
- Contact us for information regarding our sample policy
- Carton Box and Drum
- Western Europe Australia North America South America Eastern Europe Middle East Central America Asia Africa
- All India
Product Description
2-Hydroxy-3-carboxy naphthalene is a chemical compound with the molecular formula C11H6O4. It is commonly known as 2-hydroxy-3-naphthoic acid or 2-hydroxy-3-carboxynaphthalene-1-carboxylic acid. This compound is a derivative of naphthalene, a polycyclic aromatic hydrocarbon.
The systematic name for this compound is 2-hydroxy-3-carboxynaphthalene-1-carboxylic acid, and it can be abbreviated as 2-H3CNA. It belongs to the class of organic compounds known as naphthalene carboxylic acids and derivatives.
2-Hydroxy 3-Carboxy Naphthalene Properties:
1. Chemical Structure:
The compound has a naphthalene backbone with hydroxyl (-OH) and carboxyl (-COOH) functional groups. The presence of these functional groups can influence its chemical and physical properties.
2. Solubility:
The solubility of organic compounds can depend on their structure. Compounds with polar functional groups like hydroxyl and carboxyl groups are often soluble in polar solvents (e.g., water) due to the ability to form hydrogen bonds.
3. Acid-Base Properties:
The carboxyl group in the compound suggests that it can act as an acid, donating a proton (H+) in aqueous solutions.
4. Color and UV Absorption:
Naphthalene derivatives can exhibit color, and the compound may have a characteristic color. The absorption of ultraviolet (UV) light can provide information about the compounds electronic structure.
5. Biological Activity:
Depending on its structure, the compound might have biological activities or applications. Naphthalene derivatives are sometimes used in the synthesis of pharmaceuticals and other bioactive compounds.
FAQs of 2-Hydroxy 3-Carboxy Naphthalene:
Q: What is the melting point of 2-Hydroxy 3-Carboxy Naphthalene?
A: The melting point of 2-Hydroxy 3-Carboxy Naphthalene is 142-146 degree centigrade.Q: What is the boiling point of 2-Hydroxy 3-Carboxy Naphthalene?
A: The boiling point of 2-Hydroxy 3-Carboxy Naphthalene is 238-239 degree centigrade.Q: What is the taste of 2-Hydroxy 3-Carboxy Naphthalene?
A: 2-Hydroxy 3-Carboxy Naphthalene is odorless.Q: What is the physical form of 2-Hydroxy 3-Carboxy Naphthalene?
A: 2-Hydroxy 3-Carboxy Naphthalene is in liquid form.Q: How should 2-Hydroxy 3-Carboxy Naphthalene be stored?
A: 2-Hydroxy 3-Carboxy Naphthalene should be stored at room temperature.Q: What is the grade of 2-Hydroxy 3-Carboxy Naphthalene?
A: 2-Hydroxy 3-Carboxy Naphthalene is medicine grade.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Pharma Intermediates' category
 |
PRAVIN DYECHEM PVT. LTD.
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese